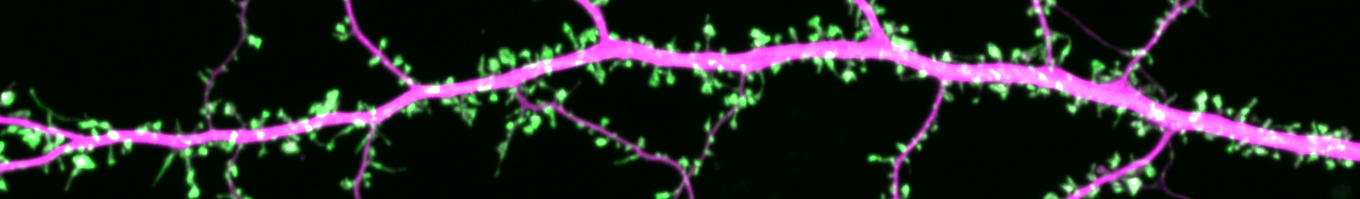
We utilize a variety of imaging techniques to visualize the incredibly dynamic protein organization in synapses of live neurons with the highest spatial and temporal resolution.
Currently, we are pouring effort into development of a new super-resolution technique called PALM, which derives its incredible power from imaging and tracking the motion of single molecules in the cell. We worked first with one of PALM’s inventors Eric Betzig and his postdoc Hari Shroff (now Chief of the Section on High Resolution Optical Imaging at the NIH) to establish conditions for executing PALM in neurons. Subsequently, we have built two customized PALM systems in the lab, and use these for day-to-day experiments. Right now, our state of the art is 2-color 3-dimensional imaging, with XY resolution better than 20 nm and axial resolution about 50 nm. We combine PALM with patch-clamp electrophysiology, Ca imaging, and other traditional modalities for deeper insight to the relationship between synapse structure and function.

We also rely on an spinning disk confocal in the lab, as well as point-scanning confocals in the School of Medicine Core Microscopy Facility, and use these for time-lapse imaging, FRAP, and photoactivation analysis of protein dynamics in synapses.
To bring our imaging approach to intact systems, we spearheaded the purchase of a new in vivo multiphoton microscope to be housed in the Core Facility. Keep an eye out as the results start rolling in, as we begin to span the gap between molecules and the mammalian brain.
We thank Andor Technology, Olympus, Zeiss, Chroma, Semrock, and others for their outstanding hardware essential for our experiments.